The statement that "runoff pH is useless" is going around RIU. It seems to be a statement which clearly states 100% lack of usefulness.
I have an open mind about runoff pH. While I don't use Hydrobuddy, I do use a spreadsheet and make my nutes from scratch. It's a PITA, tedious and time consuming, and requires concentration to avoid mistakes. I've been able to successfully alter runoff pH by altering the nute's
nitrate:ammonium ratio. I didn't discover this, instead read about it. I thought it common knowledge that ammonium sulfate (AMS) was acidifying, but maybe that knowledge was due to my lawn-care background. Manipulating the ratio of nitrate:ammonium in my nute formulas seems to change runoff pH in predictable ways. Therefore, the statement "runoff pH is useless" is false. You can certainly argue its usefulness, but useless means it lacks any usefulness.
For those who buy their nutes in easy-to-use nutrient bottles and who are not trying to change the manufacturer's designed nitrate:ammonium ratio, maybe tracking runoff pH is useless. I don't know. Here's one RIU post which may explain the rationale behind the statement "runoff pH is useless":
Hello everyone! I'm into my 2nd grow and have some issues with the measuring of pH-value. I can't understand whether to measure it directly from the soil or to measure the run-off water. I have tried both and get different values, and I'm not sure which one to go by. Because there are...
www.rollitup.org
The poster said that a soil slurry test is preferable to runoff pH. Maybe it is, for measuring potting mix pH. Is that the same thing as runoff pH? Taking a small core of potting mix which has a plant actively growing in it will likely damage some roots.
The scientists who wrote the following report used runoff pH as a data point:
Not found
journals.ashs.org
It is my belief that since I can alter runoff pH by altering the nitrate:ammonium ratio, I should set runoff to a pH value which cannabis plants are known to like and where good mineral absorptions occur. Will it make any difference in the overall performance of my grows? I don't know.
For those who believe "runoff pH is useless," would you mind pointing to any peer-reviewed journal reports which make that assertion?
 BAFA678B-4970-4FD2-AC1E-4ACFC36BA5F8.png215.8 KB · Views: 14
BAFA678B-4970-4FD2-AC1E-4ACFC36BA5F8.png215.8 KB · Views: 14 109B9BCC-FD61-4C59-806C-5F748A2A4323.png184.3 KB · Views: 14
109B9BCC-FD61-4C59-806C-5F748A2A4323.png184.3 KB · Views: 14 BA6A62FC-0F46-43CD-A574-6498EB89D395.png188 KB · Views: 9
BA6A62FC-0F46-43CD-A574-6498EB89D395.png188 KB · Views: 9 ECA380BC-6656-4184-A950-8F6AB7982C2D.png159 KB · Views: 8
ECA380BC-6656-4184-A950-8F6AB7982C2D.png159 KB · Views: 8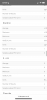 BDE73922-1B81-4476-8600-A3FE06995315.png155.2 KB · Views: 11
BDE73922-1B81-4476-8600-A3FE06995315.png155.2 KB · Views: 11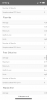 28BAE1CB-F1FA-45AA-BAFB-AE0461A17978.png158.4 KB · Views: 11
28BAE1CB-F1FA-45AA-BAFB-AE0461A17978.png158.4 KB · Views: 11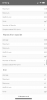 C12006AC-C702-4DD5-9A93-660128823D50.png155.4 KB · Views: 9
C12006AC-C702-4DD5-9A93-660128823D50.png155.4 KB · Views: 9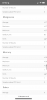 19413B5A-4B1A-4B0D-9BC3-B9ADF4ACC249.png156.2 KB · Views: 6
19413B5A-4B1A-4B0D-9BC3-B9ADF4ACC249.png156.2 KB · Views: 6 B18F2466-9AFA-44F3-BF29-1B12A5E83D7E.png151.9 KB · Views: 5
B18F2466-9AFA-44F3-BF29-1B12A5E83D7E.png151.9 KB · Views: 5 A4991F91-DBFA-4D5F-B227-368AFC48E84F.png154.9 KB · Views: 4
A4991F91-DBFA-4D5F-B227-368AFC48E84F.png154.9 KB · Views: 4

